New research indicates that some cases of Alzheimer’s disease may involve infection with Porphyromonas gingivalis, a bacterial species that is best known for causing gum disease in the mouth.
A new treatment for Alzheimer’s disease may already be underway – and it is targeting the microbes found in the mouth.
A study published today in the journal Science Advances points to an unlikely villain in neurodegenerative disease: Porphyromonas gingivalis, a bacterium that causes gum disease, may be able to travel from the mouth to the brain, where it can induce chemical changes that damage cognition. These findings suggest that some cases of Alzheimer’s disease may be infectious, and a new drug that blocks this process is currently in clinical trials.
“It’s very exciting in many ways,” says Sim Singhrao, who studies neurodegenerative disease at the University of Central Lancashire in the UK but was not involved in the new study. “This work not only highlights the problem of poor oral hygiene as a risk factor for Alzheimer’s disease, but may also offer a futuristic solution.”
Periodontitis and Alzheimer’s disease.
P. gingivalis is best known for causing periodontitis, an inflammatory disease that constantly eats away at the gums and bones that support the teeth, in part because the gum’s microorganism produces powerful enzymes that chew other proteins. But in recent years, scientists have begun to cautiously link Alzheimer’s disease to P. gingivalis infection – pointing to the possibility that these mouth-loving microbes may be able to open a store in the brain.
In a new study, a team led by Stephen Domina and Casey Lynch of Cortexyme, a San Francisco-based company developing Alzheimer’s therapies, found bacterial gingipains in the brains of over 90 percent of a group of more than 50 Alzheimer’s patients who died. The more gingipains an individual has, the more the brain shows chemical signs of deterioration, including the build-up of a protein called tau. Under normal circumstances, tau helps neurons maintain structural integrity. But in brains progressing to Alzheimer’s disease, this protein can undergo abnormal chemical changes and become entangled in nodes inside the brain cells, deteriorating their ability to send signals to one another.
Porphyromonas gingivalis, a species of bacteria known to cause gum disease, may be capable of infiltrating the brain. There, its presence can trigger an immune response that ultimately impairs the function of brain cells.
Probable mechanism
When scientists introduced P. gingivalis into the mouths of eight mice, they found that the bacteria migrated to all of the brains within a few weeks. In response to the infection, the mice also seemed to increase their production of a protein called beta amyloid. Like tau, amyloid beta occurs naturally in the brain but behaves abnormally in Alzheimer’s patients by forming sticky aggregates outside of the cells.
What amyloid beta does under normal circumstances is not well understood. But some researchers, including Dominy and Lynch, believe that it plays a role in the immune system and is produced when the body senses an unwanted microbe. “It’s a bit like the body is sending out a little mesh saying,” I’m going to trap these bacteria, “says Singhrao.
As good as it may sound, the protective abilities of beta amyloid can ultimately cause harm. When P. gingivalis infiltrates the brain, amyloid beta can be channeled into overdrive – and if these microbial trapping networks are not removed, they can stick together, blocking communication lines between brain cells. As with tau, these cell blockages can eventually push the brain towards dementia.
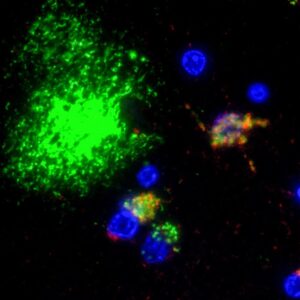 But Dominy believes there is more to this than an overactive immune system. “We believe the guilty are really the culprits,” he says. “They directly damage the brain.”
But Dominy believes there is more to this than an overactive immune system. “We believe the guilty are really the culprits,” he says. “They directly damage the brain.”
Accordingly, when scientists exposed the mice to a strain of P. gingivalis that did not have its destructive gingipains, the rodents suffered from minor Alzheimer’s-like lesions in the brain. And when P. gingivalis-infected mice were treated with the new anti-gingival drug Cortexyme, fewer bacteria attacked the brain and less amyloid beta accumulation was.
Cortexyme is already in human clinical trials, and Lynch says the early results are encouraging. Regardless of the outcome for this particular drug, the Alzheimer’s case is far from over. Stronger links between oral infections and Alzheimer’s disease still need to be established in humans, says Allison Reiss, an Alzheimer researcher at Winthrop University in New York, who was not involved in the new study. After all, he says, while the results of the mice studies are encouraging, what happens in rodents isn’t always summarized in humans.
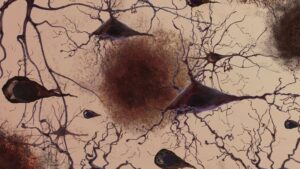
Plaque that accumulates between neurons is the hallmark of Alzheimer’s disease.
Ming Guo, a neuroscientist and Alzheimer expert at the University of California, Brain Research Institute in Los Angeles, who was not involved in the new study, even though the results work for human populations, are not all the same. You may find that bacterial infections are only one possible route of dementia and mental retardation to develop in an extremely complex disease.
“Alzheimer’s is a complex disorder, not a single complex point,” says Reiss.
Many other questions remain. It’s still not entirely clear how P. gingivalis breaches brain barriers in mice or humans – or how risky it is for these bacteria to live in you in the first place. Numbers vary, but P. gingivalis can live in the mouths of up to 40 percent of healthy people, and many of these people will not experience negative effects.
In other words, P. gingivalis infection does not guarantee Alzheimer’s disease, and there is no conclusive evidence that all Alzheimer’s cases originate from P. gingivalis. Regardless, Singhrao says oral hygiene won’t hurt – if not for your mind, then at least for your mouth.


